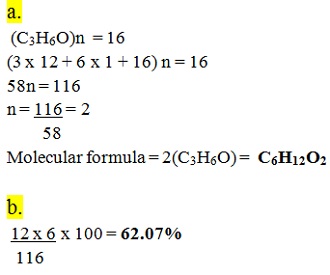Question
A compound has an empirical formula C3H6O and a relative formula mass of 116.
a. Determine its molecular formula (H = 1.0, C = 12.0, O = 16.0)
b. Calculate the percentage composition of carbon by mass in the compound.
a. Determine its molecular formula (H = 1.0, C = 12.0, O = 16.0)
b. Calculate the percentage composition of carbon by mass in the compound.
Answer